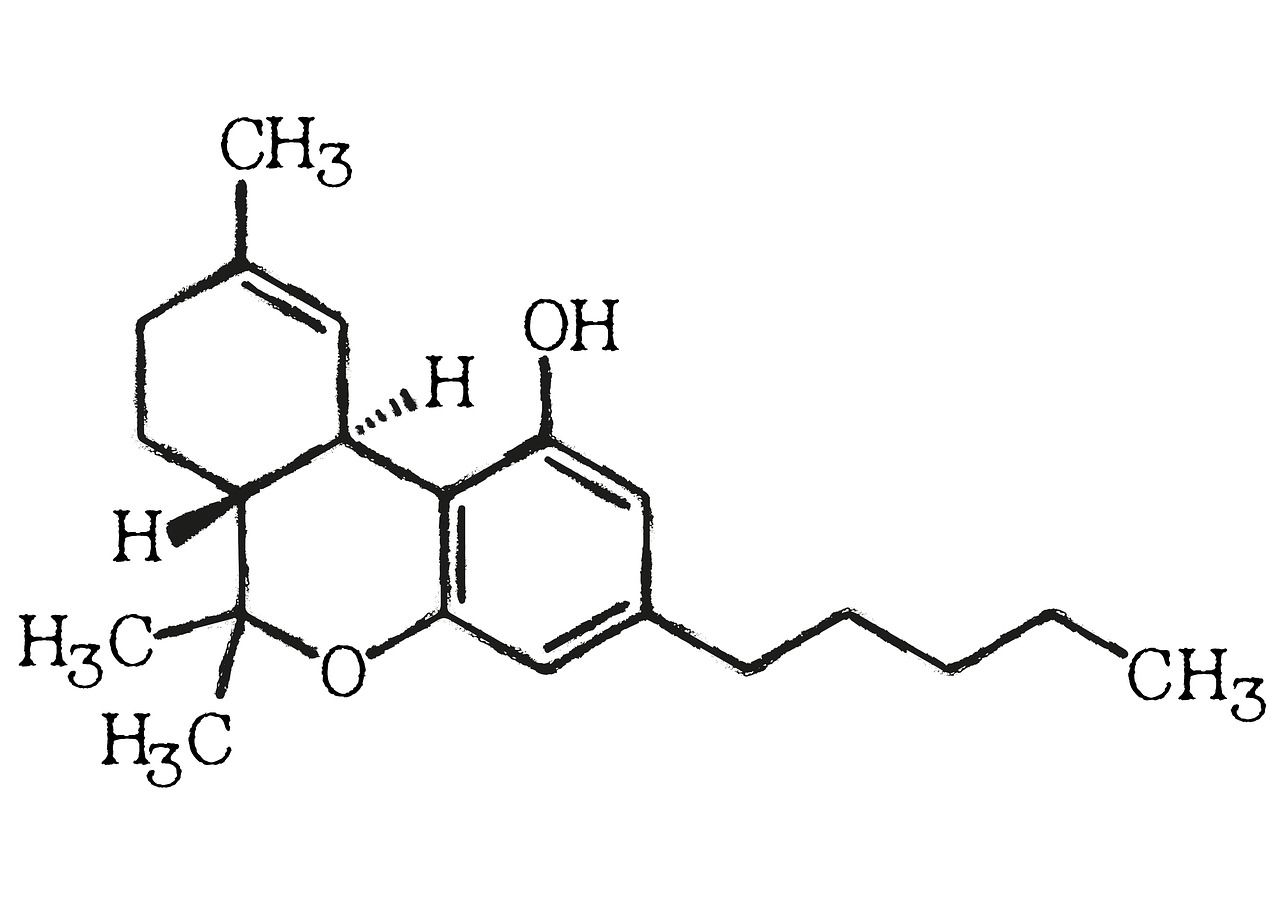
CBD built the first big tent for hemp wellness, but the next wave of differentiation will come from rare cannabinoids—compounds present in trace amounts that enable novel product architectures and claims. One standout is cannabicyclol (CBL), a non-intoxicating cannabinoid formed when cannabichromene (CBC) rearranges under light or heat—an origin story that matters for sourcing and manufacturing.
From a product-development lens, CBL invites “function-forward” formulations that go beyond CBD-only positioning. Early reviews classify CBL as non-psychoactive—useful for brands seeking calm-or-clear products without an intoxicating effect profile. While robust human data are still scarce, authoritative reviews note no known psychoactivity for CBL and CBC, signaling potential for daytime or focus-oriented lines where consumer acceptance hinges on clarity and non-impairment.
Supply will define strategy. Because CBL arises mainly through photochemical conversion of CBC, brands can pursue two routes: (1) work with aged or specifically treated biomass that favors CBL formation, or (2) isolate CBC and convert it under controlled conditions prior to purification. Recent analytical work on CBC/CBL separations underscores that dedicated chromatography and characterization are required—an operational reminder that genuine CBL SKUs will rely on specialist processing and validated testing.
Market signals suggest the rare-cannabinoid category is material, not niche. U.S. “minor cannabinoid” sales (a basket that includes CBG, CBN, THCV, and others) are already measured in the billions and forecast double-digit CAGR through the decade, creating shelf room for SKUs that spotlight specific effects or use cases, rather than generic “CBD wellness.” For retailers, that translates to new price ladders and premium tiers; for manufacturers, it argues for platform formulations where CBD is the chassis and rare cannabinoids like CBL are tunable modules.
Quality will be the moat. Independent investigations continue to find mislabeling in mainstream CBD products; layering rarer analytes only raises the bar for validated methods and transparent Certificates of Analysis (COAs). Product teams should require third-party labs to quantify CBL alongside CBD/THC, report method limits of detection, and screen for solvents and contaminants—critical to avoid recreating CBD’s accuracy problems at a higher price point.
Regulatory reality is another pivot point. Under the 2018 Farm Bill, hemp derivatives under 0.3% Δ-9-THC are federally legal as “hemp,” but FDA has not recognized CBD (let alone rare cannabinoids) as lawful dietary supplements or conventional foods; warning letters continue for impermissible claims and noncompliant formats. Meanwhile, Congress has floated amendments that could significantly narrow what hemp-derived cannabinoids can be sold—policies aimed largely at intoxicating derivatives, but with potential spillover effects on the broader category. Product roadmaps should be scenario-planned for both a tighter federal definition and continued state-by-state divergence.
What should brands do now?
- Own the chemistry. If a SKU claims CBL, document the CBC-to-CBL conversion, purification, and stability data, then translate that into consumer-friendly education.
- Design for verification. Publish batch COAs that include CBL quantitation and contaminants panels; commit to label accuracy tolerances.
- Build effect-driven stacks. Pair CBD with CBL (and possibly CBC/CBG) to target distinct need states—calm, recovery, focus—supported by conservative, non-disease claims while research matures.
- Future-proof compliance. Track FDA actions and Farm Bill updates; maintain state-specific labels and distribution rules to avoid sudden delistings.
Bottom line: rare cannabinoids like CBL won’t replace CBD—they’ll refine it. The brands that win will treat CBL not as a buzzword, but as a transparently sourced, analytically verified ingredient that unlocks clearer product roles, premium pricing, and consumer trust.
Read what the future of CBD in the U.S. marketplace holds here.