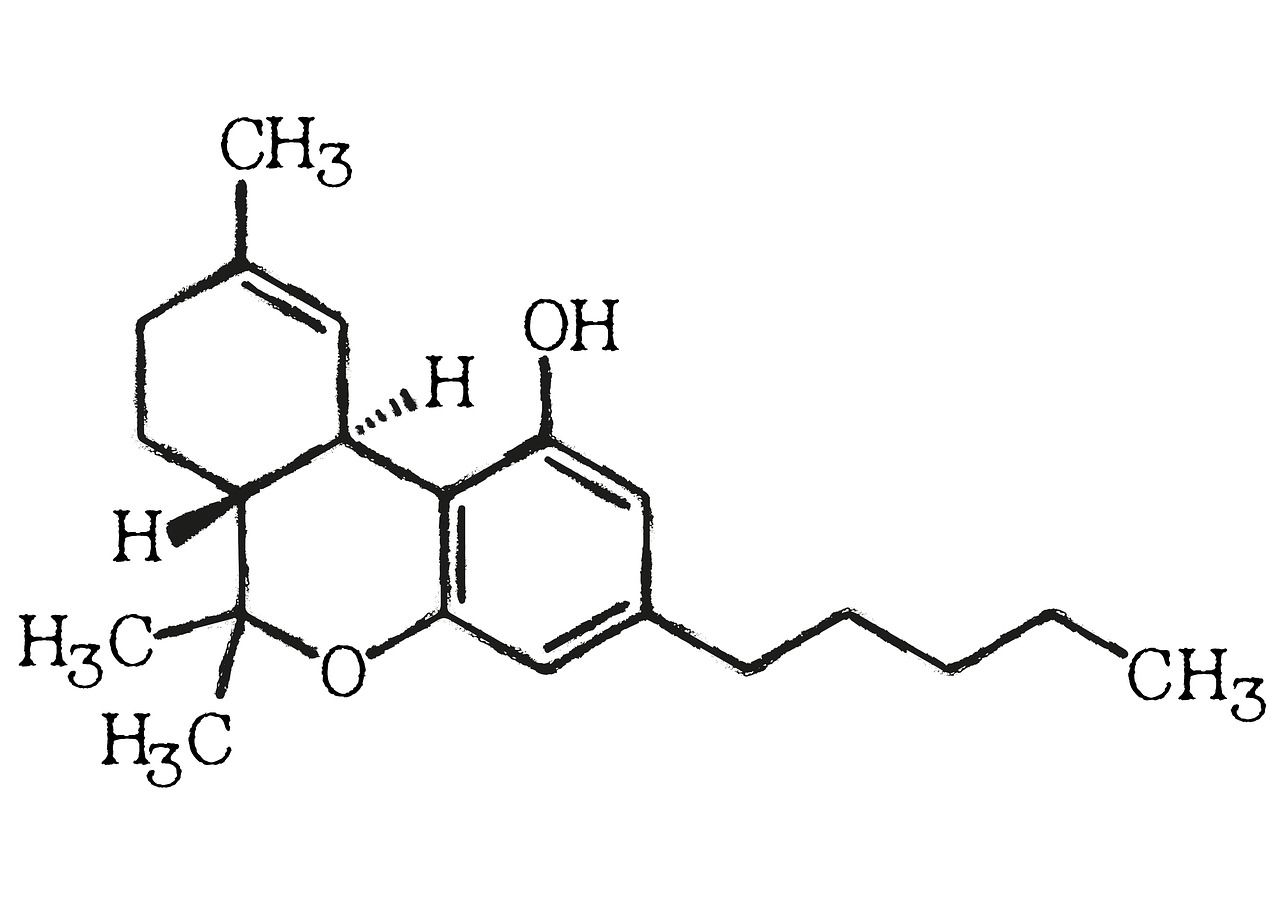
Understanding Cannabicyclol: Its Formation and Stability in Cannabis
Cannabicyclol (CBL) is one of the lesser-known cannabinoids present in the cannabis plant.
What Makes CBL Non-Psychoactive—and Why That Matters to Users
As science continues to uncover the secrets of cannabis, lesser-known cannabinoids like cannabicyclol (CBL) are stepping into the spotlight.
How CBL Interacts with CB1 and CB2 Receptors
The human endocannabinoid system (ECS) regulates a wide range of physiological functions, including mood, pain perception, appetite, memory, and immune response.